Ecological site group R023XY909OR
Clayey Mesic Plateaus 8-14 PZ Wyoming Big Sagebrush and Thurber's Needlegrass
Last updated: 06/03/2024
Accessed: 04/06/2026
Ecological site group description
Key Characteristics
- Site does not pond or flood
- Landform other than dunes
- Surface soils are not clayey
- Sites are shrub or grass dominated
- MAP ≤ 10"
- Texture (PCS) ashy or loamy
Provisional. A provisional ecological site description has undergone quality control and quality assurance review. It contains a working state and transition model and enough information to identify the ecological site.
Physiography
This group is on plateaus across the MLRA at elevations between 4,000 and 7,000 feet. Slopes are 1 to 65 percent. Slopes from 5 to 25 percent are most common.
Climate
The climate is classified as Cold Semi-Arid in the Koppen Classification System.
The area receives 8 to 14 inches of annual precipitation as snow in the winter and rain in spring and fall. Summers are generally dry.
The frost-free period is 65 to 120 days. The mean annual air temperature is 42 to 54 °F.
Soil features
There is significant variability in soil depth and surface texture between sites in this group. Though variable, soil textures are predominantly clayey or ashy. Parent materials are alluvium and colluvium.
Taxonomically, this group is divided between Mollisols and Aridisols.
Common soil series include Bucklake, Old Camp, Reywat, and Saraph.
Vegetation dynamics
Ecological Dynamics and Disturbance Response:
An ecological site is the product of all the environmental factors responsible for its development. Each site has a set of key characteristics that influence its resilience to disturbance and resistance to invasives. According to Caudle et al. (2013), key characteristics include:
1. Climate factors such as precipitation and temperature.
2. Topographic characteristics such as aspect, slope, elevation, and landform.
3. Hydrologic processes such as infiltration and runoff.
4. Soil characteristics such as depth, texture, structure, and organic matter.
5. Plant communities and their functional groups and productivity.
6. Natural disturbance (fire, herbivory, etc.) regime.
Biotic factors that influence resilience include site productivity, species composition and structure, and population regulation and regeneration (Chambers et al., 2013).
The ecological sites in this group are dominated by deep-rooted, cool-season, perennial bunchgrasses and long-lived shrubs (at least 50 years old) with high root to shoot ratios. The dominant shrubs usually root to the full depth of the winter-spring soil moisture recharge, which ranges from 1.0 to over 3.0 meters (Comstock & Ehleringer, 1992). Root length of mature sagebrush plants was measured to a depth of 2 meters in alluvial soils in Utah (Richards & Caldwell, 1987). These shrubs have a flexible generalized root system with development of both deep taproots and laterals near the surface (Dobrowolski et al., 1990).
In the Great Basin, most of the annual precipitation occurs during the winter and early spring. This continental semiarid climate regime favors growth and development of deep-rooted shrubs and herbaceous cool season plants using the C3 photosynthetic pathway (Comstock & Ehleringer, 1992). Winter precipitation and slow melting of snow results in deeper percolation of moisture into the soil profile. Herbaceous plants, more shallow-rooted than shrubs, grow earlier in the growing season and thrive on spring rains, while the deeper-rooted shrubs lag in phenological development because they draw from deeply infiltrating moisture from snowmelt the previous winter. Periodic drought regularly influences sagebrush ecosystems, and drought duration and severity have increased throughout the 20th century in much of the Intermountain West. Major shifts away from historical precipitation patterns have the greatest potential to alter ecosystem function and productivity. The timing of precipitation and water availability within the soil profile can alter species composition and productivity (Bates et al., 2006).
Variability in plant community composition and production depends on soil surface texture and depth. Thurber’s needlegrass (Achnatherum thurberianum) increases on gravelly soils, whereas Indian ricegrass (Achnatherum hymenoides) increases on sandy soil surfaces, and squirreltail (Elymus elymoides) increases on silty soil surfaces. A weak argillic horizon promotes production of bluebunch wheatgrass (Pseudoroegneria spicata). Production generally increases with soil depth. The amount of sagebrush in the plant community depends on disturbances like fire, Aroga moth infestations, and grazing. Sandberg bluegrass (Poa secunda) more easily dominates sites where surface soils are gravelly loams or where there is an increase in ash in the upper soil profile.
Wyoming big sagebrush (Artemisia tridentata ssp. wyomingensis) is the most drought-tolerant of the big sagebrush subspecies. It is generally long-lived. Therefore, it is not necessary for new individuals to recruit every year for perpetuation of the stand. Infrequent, large recruitment events and simultaneous low, continuous recruitment are the foundation of population maintenance (Noy-Meir, 1973). Survival of the seedlings depends on adequate moisture conditions.
Native insect outbreaks are also important drivers of ecosystem dynamics in sagebrush communities. Climate is generally believed to influence the timing of insect outbreaks, especially outbreaks of a sagebrush defoliator called Aroga moth (Aroga websteri). Aroga moth infestations occurred in the Great Basin in the 1960s, the early 1970s, and have been ongoing in Nevada since 2004 (Bentz et al., 2008). Thousands of acres of big sagebrush have been impacted, with partial to complete die-off observed. The Aroga moth can partially or entirely kill individual plants or entire stands of big sagebrush (Furniss & Barr, 1975).
Bunchgrasses generally have somewhat shallower root systems than the shrubs on these sites; root densities of bunchgrasses are often as high as or higher than those of shrubs in the upper 0.5 meters of the soil profile. The general differences in root depth distributions between grasses and shrubs result in resource partitioning in these shrub/grass systems.
The Great Basin sagebrush communities have high spatial and temporal variability in precipitation both among years and within growing seasons (MacMahon, 1980). Nutrient availability is typically low but increases with elevation and closely follows moisture availability. The invasibility of plant communities is often linked to resource availability. Disturbance changes resource uptake and increases nutrient availability, often to the benefit of non-native species; native species are often damaged and their ability to use resources is depressed for a time and resource pools may increase from lack of use and/or the decomposition of dead plant material following disturbance (Whisenant, 1999; Miller et al., 2013). The invasion of sagebrush communities by cheatgrass (Bromus tectorum) is linked to disturbances (fire, abusive grazing) that result in fluctuations in resources (Beckstead & Augspurger, 2004; Chambers et al., 2007; Johnson et al., 2011).
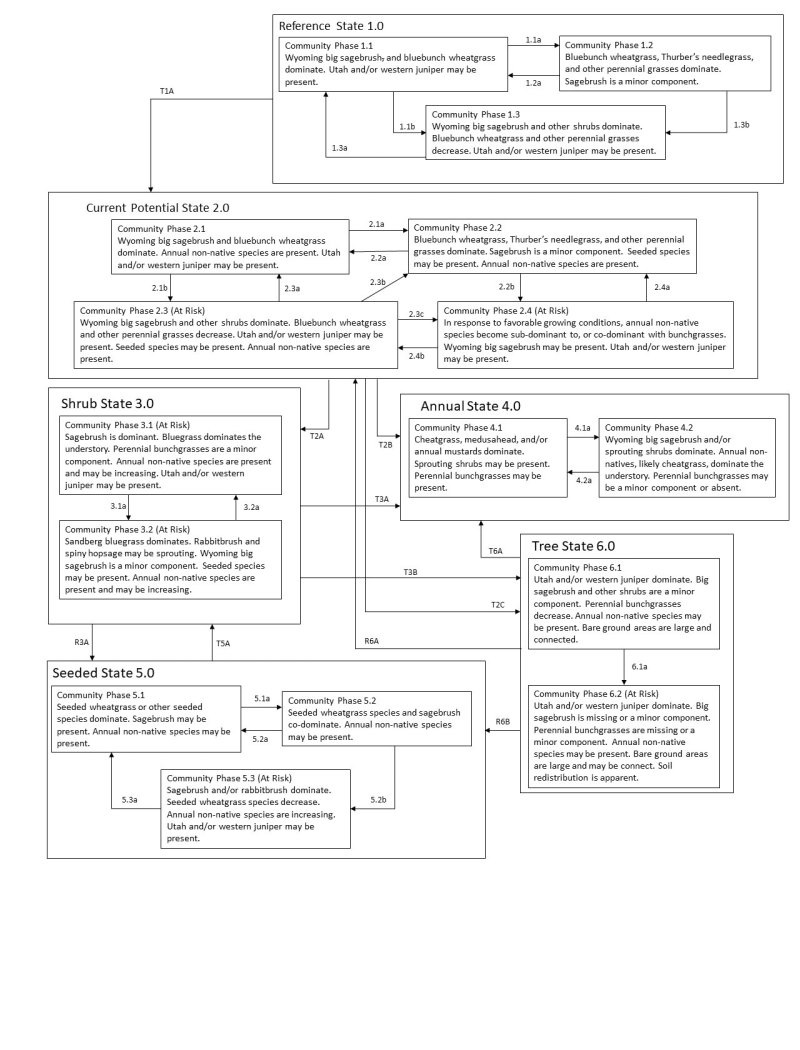
The introduction of annual weedy species, like cheatgrass, may increase fire frequency and eventually lead to an annual state. Conversely, as fire frequency decreases, sagebrush will increase. When combined with inappropriate grazing management, the perennial bunchgrasses and forbs may decrease.
At the upper end of this group’s precipitation range, there is potential for infilling by Utah juniper (Juniperus osteosperma). Infilling may also occur on sites adjacent to woodland sites or other ecological sites where juniper is present. Without disturbance in these areas, Utah juniper will eventually dominate the site and out-compete sagebrush for water and sunlight, thus severely reducing both the shrub and herbaceous understory (Miller & Tausch, 2001; Lett & Knapp, 2005). The potential for soil erosion increases as the woodland matures and the understory plant community cover declines (Pierson et al., 2010).
The ecological sites in this group have low resilience to disturbance and low resistance to invasion. Resilience increases with elevation, northerly aspect, precipitation, and nutrient availability.
Five possible stable states have been identified for the Loamy 8-10” ecological site. Differences in resilience to disturbance for the remaining ecological sites in this group are described at the end of this document.
Annual Invasive Grasses:
The species most likely to invade these sites are cheatgrass and medusahead (Taeniatherum). Both species are cool- season annual grasses that maintain an advantage over native plants in part because they are prolific seed producers, able to germinate in the autumn or spring, tolerant of grazing and increase with frequent fire (Klemmedson & Smith, 1964; Miller et al., 1999). Medusahead and cheatgrass originated from Eurasia and both were first reported in North America in the late 1800s (Mack & Pyke, 1983; Furbush, 1953). Pellant and Hall (1994) found 3.3 million acres of public lands dominated by cheatgrass and suggested that another 76 million acres were susceptible to invasion by winter annuals including cheatgrass and medusahead. By 2003, medusahead occupied approximately 2.3 million acres in 17 western states (Rice, 2005). In the Intermountain West, the exponential increase in dominance by medusahead has largely been at the expense of cheatgrass (Harris, 1967; Hironaka, 1994). Medusahead matures two to three weeks later than cheatgrass (Harris, 1967). Recently, James et al. (2008) measured leaf biomass over the growing season and found that medusahead maintained vegetative growth later in the growing season than cheatgrass. Mangla et al. (2011) also found medusahead had a longer period of growth and more total biomass than cheatgrass and hypothesized this difference in relative growth rate may be due to the ability of medusahead to maintain water uptake as upper soils dry compared to co-occurring species, especially cheatgrass. Medusahead litter has a slow decomposition rate because of its high silica content, allowing it to accumulate over time and suppress competing vegetation (Bovey et al., 1961; Davies & Johnson, 2008). Harris (1967) reported medusahead roots have thicker cell walls compared to those of cheatgrass, allowing it to more effectively conduct water, even in very dry conditions.
Recent modeling and empirical work by Bradford and Lauenroth (2006) suggest that seasonal patterns of precipitation input and temperature are also key factors determining regional variation in the growth, seed production, and spread of invasive annual grasses. Collectively, the body of research suggests that the invasion and dominance of medusahead onto native grasslands and cheatgrass-infested grasslands will continue to increase in severity because conditions that favor native bunchgrasses or cheatgrass over medusahead are rare (Mangla et al., 2011). Medusahead replaces native vegetation and cheatgrass directly by competition and suppression. It replaces native vegetation indirectly by increasing fire frequency.
Methods to control medusahead and cheatgrass include herbicide, fire, grazing, and seeding of primarily non-native wheatgrasses. Mapping potential or current invasion vectors is a management method designed to increase the cost effectiveness of control methods. A study by Davies et al. (2013) found an increase in medusahead cover near roads. Cover was higher near animal trails than random transects, but the difference was less evident. This implies that vehicles and animals aid the spread of the weed; however, vehicles are the major vector of movement. Spraying with herbicide (Imazapic or Imazapic + glyphosate) and seeding with crested wheatgrass (Agropyron cristatum) and Sandberg bluegrass have been more successful at combating medusahead and cheatgrass than spraying alone (Sheley et al., 2012). Where native bunchgrasses are missing from the site, revegetation of medusahead- or cheatgrass-invaded rangelands has shown a higher likelihood of success when using introduced perennial bunchgrasses such as crested wheatgrass (Davies et al., 2015). Butler et al. (2011) tested four herbicides (Imazapic, Imazapic + glyphosate, rimsulfuron, and sulfometuron + Chlorsulfuron), using herbicide-only treatments, for suppression of cheatgrass, medusahead, and ventenata (Ventenata dubia) within residual stands of native bunchgrass. Additionally, they tested the same four herbicides followed by seeding of six bunchgrasses (native and non-native) with varying success. Herbicide-only treatments appeared to remove competition for established bluebunch wheatgrass by providing 100 percent control of ventenata and medusahead and greater than 95 percent control of cheatgrass. However, caution in using these results is advised, as only one year of data was reported.
Prescribed fire has also been utilized in combination with the application of pre-emergent herbicide to control medusahead and cheatgrass (J. L. Vollmer & J. G. Vollmer, 2008). Mature medusahead or cheatgrass is very flammable and fire can be used to remove the thatch layer, consume standing vegetation, and even reduce seed levels. Furbush (1953) reported that timing a burn while the seeds were in the milk stage effectively reduced medusahead the following year. He further reported that adjacent unburned areas became a seed source for reinvasion the following year.
When considering the combination of pre-emergent herbicide and prescribed fire for invasive annual grass control, it is important to assess the tolerance of desirable brush species to the herbicide being applied. J. L. Vollmer and J. G. Vollmer (2008) tested the tolerance of mountain mahogany (Cercocarpus montanus), antelope bitterbrush (Purshia tridentata), and multiple sagebrush species to three rates of Imazapic and the same rates with methylated seed oil as a surfactant. They found a cheatgrass control program in an antelope bitterbrush community should not exceed Imazapic at 8 ounces per acre with or without surfactant. Sagebrush, regardless of species or rate of application, was not affected. However, many environmental variables were not reported in this study and managers should install test plots before broad scale herbicide application is initiated.
Fire Ecology:
Wyoming big sagebrush communities historically had low fuel loads. Patchy fires that burned in a mosaic pattern were common at 10- to 70-year fire return intervals (Young et al., 1979; West & Hassan, 1985; Bunting et al., 1987). However, newer studies suggest longer return intervals. Davies et al. (2006) suggest fire return intervals in Wyoming big sagebrush communities were around 50 to 100 years. More recently, Baker (2011) estimates fire return intervals were 200 to 350 years in Wyoming big sagebrush communities. Wyoming big sagebrush is killed by fire and only regenerates from seed. Recovery time for Wyoming big sagebrush may require 50 to 120 or more years (Baker, 2006). However, the introduction and expansion of cheatgrass has dramatically altered the fire regime (Balch et al., 2013) and restoration potential of Wyoming big sagebrush communities.
The effect of fire on bunchgrasses relates to culm density, culm-leaf morphology, and the size of the plant. The initial condition of bunchgrasses on a site and seasonality and intensity of the fire all factor into the individual species response. For most forbs and grasses, the growing points are located at or below the soil surface. This provides relative protection from disturbances that remove above ground biomass, such as grazing or fire. Thus, grass mortality after fire relates directly to culm density, culm-leaf morphology, size of plant, and abundance of old growth because these factors increase duration and intensity of heat at the plant base (Wright, 1971; Young, 1983).
Burning has been found to decrease the vegetative and reproductive vigor of Thurber’s needlegrass (Uresk et al., 1976). Fire can cause high mortality and reduce basal area and yield of Thurber’s needlegrass (Britton et al., 1990). The fine leaves and densely tufted growth form make this grass susceptible to subsurface charring of the crowns (Wright & Klemmedson, 1965). Although timing of fire highly influences the response and mortality of Thurber’s needlegrass, smaller bunch sizes are less likely to be damaged by fire (Wright & Klemmedson, 1965). Thurber’s needlegrass often survives fire and continues growth or regenerates from tillers when conditions are favorable (Koniak, 1985; Britton et al., 1990). Reestablishment on burned sites is relatively slow due to low germination and competitive ability (Koniak, 1985). Cheatgrass is a highly successful competitor with seedlings of this needlegrass and may preclude reestablishment (Evans & Young, 1978).
Indian ricegrass is fairly fire-tolerant (Wright, 1985) likely due to its low culm density and below ground plant crowns. Indian ricegrass reestablishes on burned sites via seed dispersed from adjacent unburned areas (Young, 1983; West, 1994). Thus, the presence of surviving, seed-producing plants is necessary for reestablishment of Indian ricegrass. It is important to manage grazing following fire to promote seed production and establishment of seedlings.
Fire will remove aboveground biomass from bluebunch wheatgrass, but plant mortality is generally low (Robberecht & Defossé, 1995) because the buds are underground (Conrad & Poulton, 1966) or protected by foliage. Uresk et al. (1976) reported burning increased vegetative and reproductive vigor of bluebunch wheatgrass. Thus, bluebunch wheatgrass experiences slight damage from fire but is more susceptible to fire damage in drought years (Young, 1983). Plant response varies depending on season, fire severity, fire intensity, and post-fire soil moisture availability.
Sandberg bluegrass, a minor component of these ecological sites, can increase following fire likely due to its low stature and productivity (Daubenmire, 1975). Sandberg bluegrass may impair reestablishment of deeper-rooted bunchgrasses. Reduced bunchgrass vigor or density provides an opportunity for Sandberg bluegrass expansion and/or cheatgrass and other invasive species to occupy interspaces, leading to increased fire frequency, and potentially an annual plant community.
The range and density of Utah juniper has increased since the middle of the nineteenth century (Tausch, 1999; Miller & Tausch, 2001). The expansion of trees into sagebrush ecosystems is caused by wildfire suppression, historic livestock grazing, and climate change (Bunting, 1994).
Depending on fire severity, rabbitbrush and horsebrush may increase after fire. Rubber rabbitbrush (Ericameria nauseosa) is top-killed by fire, but can resprout after fire and can also establish from seed (Young, 1983). Yellow rabbitbrush (Chrysothamnus viscidiflorus) is top-killed by fire, but sprouts vigorously after fire (Kuntz, 1982; Akinsoji, 1988). As cheatgrass increases, fire frequencies also increase. Some frequencies reach between 0.23 and 0.43 fires per year, at which point even sprouting shrubs such as rabbitbrush will not survive (Whisenant, 1990).
The grasses likely to invade this site are cheatgrass and medusahead. These invasive grasses displace desirable perennial grasses, reduce livestock forage, and accumulate large fuel loads that foster frequent fires (Davies & Svejcar, 2008). Invasion by annual grasses can alter the fire cycle by increasing fire size, fire season length, rate of spread, numbers of individual fires, and likelihood of fires spreading into native or managed ecosystems (D’Antonio & Vitousek, 1992; Brooks et al., 2004). While historical fire return intervals are estimated at 15 to 100 years, areas dominated by cheatgrass are estimated to have a fire return interval of 3 to 5 years (Whisenant, 1990). The mechanisms by which invasive annual grasses alter fire regimes likely interact with climate. For example, cheatgrass cover and biomass vary with climate (Chambers et al., 2007) and are promoted by wet and warm conditions during the fall and spring. Invasive annual species can take advantage of high nitrogen availability following fire because of their higher growth rates and increased seedling establishment relative to native perennial grasses (Monaco et al., 2003).
Wildlife/Livestock Grazing Interpretations:
Many wildlife species depend on the sagebrush ecosystem, such as the greater sage grouse, sage sparrow, pygmy rabbit and sagebrush vole. Dobkin and Sauder (2004) identified 61 animal species, including 24 mammals and 37 birds, associated with the shrub-steppe habitats of the Intermountain West.
Overgrazing leads to an increase in sagebrush and a decline in understory plants like Thurber’s needlegrass. Subsequently, squirreltail or Sandberg bluegrass become the dominant understory species.
Invasions of annual weedy forbs and cheatgrass can be caused by grazing degradation. Invasion leads to a decline in squirreltail and bluegrasses and an increase in bare ground. A combination of overgrazing and prolonged drought leads to soil erosion, increased bare ground, and decreased plant production.
Sites with cheatgrass present can transition to cheatgrass-dominated communities following wildfire. Without management, cheatgrass and annual forbs are likely to invade and dominate these sites, especially after fire. Although trees are not part of this ecologic site group concept, Utah juniper can invade and eventually dominate sites.
Thurber's needlegrass is an important forage source for livestock and wildlife in the arid regions of the West (Ganskopp, 1988). The seeds are apparently not injurious, but grazing animals avoid them when the seeds begin to mature. Heavy grazing during the growing season has been shown to reduce the basal area of Thurber’s needlegrass (Eckert & Spencer, 1987). This suggests that both seasonality and utilization are important factors in management of this plant. A single defoliation, particularly during the boot stage, can reduce herbage production and root mass, thus potentially lowering the competitive ability of this needlegrass (Ganskopp, 1988).
Bluebunch wheatgrass is moderately grazing-tolerant and is very sensitive to defoliation during the active growth period (Blaisdell & Pechanec, 1949; Laycock, 1967; Anderson & Scherzinger, 1975). In studies, herbage and flower stalk production were reduced with clipping at all times during the growing season; clipping was most harmful, however, during the boot stage (Blaisdell & Pechanec, 1949; Britton et al., 1990). Tiller production and growth of bluebunch wheatgrass can be greatly reduced when clipping is coupled with drought (Busso & Richards, 1995). Mueggler (1975) estimated that low-vigor bluebunch wheatgrass may need up to 8 years rest to recover. Although an important forage species, it is not always the preferred species by livestock and wildlife.
Reduced bunchgrass vigor or density provides an opportunity for Sandberg bluegrass expansion and/or cheatgrass and other invasive species—saltlover (Halogeton glomeratus), curveseed butterwort (Ceratocephala testiculata), and annual mustards—to occupy interspaces. Sandberg bluegrass increases under grazing pressure (Tisdale & Hironaka, 1981) and is capable of co-existing with cheatgrass. Excessive sheep grazing favors Sandberg bluegrass; however, where cattle are the dominant grazers, cheatgrass often dominates (Daubenmire, 1970). Thus, depending on the season of use, the type of grazing animal, and site conditions, either Sandberg bluegrass or cheatgrass may become the dominant understory species with inappropriate grazing management.
Long-term disturbance response may be influenced by small differences in landscape topography. Concave depressions hold more moisture and may retain deep-rooted perennial grasses, whereas convex areas are less resilient and may have more Sandberg bluegrass present.
Inappropriate grazing practices can be tied to the success of medusahead, but eliminating grazing will not eradicate medusahead if it is already present (Wagner et al., 2001). Sheley and Svejcar (2009) reported that even moderate defoliation of bluebunch wheatgrass resulted in increased medusahead density. They suggested that disturbances such as plant defoliation limit soil resource capture, which creates an opportunity for exploitation by medusahead. Avoidance of medusahead by grazing animals allows medusahead populations to expand. This creates seed reserves that can infest adjoining areas and cause changes to the fire regime. Medusahead replaces native vegetation and cheatgrass directly by competition and suppression and native vegetation indirectly by an increase in fire frequency.
Medusahead litter has a slow decomposition rate because of its high silica content, allowing it to accumulate over time and suppress competing vegetation (Bovey et al., 1961; Davies & Johnson, 2008).
References:
Akinsoji, A. 1988. Postfire vegetation dynamics in a sagebrush steppe in southeastern Idaho, USA.Vegetation 78(3):151-155.
Beckstead, J., and Augspurger, C. K. 2004. An experimental test of resistance to cheatgrass invasion: limiting resources at different life stages. Biological Invasions 6(4):417-432.
Bentz, B., D. Alston, and T. Evans. 2008. Great Basin Insect Outbreaks. In: J. Chambers, N. Devoe, A. Evenden (eds.). Collaborative Management and Research in the Great Basin -- Examining the issues and developing a framework for action Gen. Tech. Rep. RMRS-GTR-204. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fort Collins, CO. Pages 45-48.
Blaisdell, J. P. and J. F. Pechanec. 1949. Effects of herbage removal at various dates on vigor of bluebunch wheatgrass and arrowleaf balsamroot. Ecology 30(3):298-305.
Bovey, W. R., D. Le Tourneau, and C. L. Erickson. 1961. The chemical composition of medusahead and downy brome. Weeds 9(2):307-311.
Bradford, J. B., and W. K. Lauenroth. 2006. Controls over invasion of Bromus tectorum: The importance of climate, soil, disturbance and seed availability. Journal of Vegetation Science 17(6):693-704.
Britton, C. M., G. R. McPherson and F. A. Sneva. 1990. Effects of burning and clipping on five bunchgrasses in eastern Oregon. The Great Basin Naturalist 50(2):115-120.
Brooks, M. L., C. M. D'Antonio, D. M. Richardson, J. B. Grace, J. E. Keeley, J. M. Ditomaso, R. J. Hobbs, M. Pellant, and D. Pyke. 2004. Effects of Invasive Alien Plants on Fire Regimes. BioScience 54(7):677-688.
Bunting, S. 1994. Effects of Fire on Juniper woodland ecosystems in the Great Basin. In: S. B. Monsen and S. G. Ketchum, (eds.). Proceedings: Ecology and Management of Annual Rangelands. Gen. Tech. Rep. INT-GTR-313. 1992, May 18-21. U.S. Department of Agriculture, Forest Service, Intermountain Research Station., Boise, ID. Pages 53-55.
Butler, M., R. Simmons, and F. Brummer. 2011. Restoring Central Oregon Rangeland from Ventenata and Medusahead to a Sustainable Bunchgrass Environment – Warm Springs and Ashwood. Central Oregon Agriculture Research and Extension Center. COARC 2010. Pages 77-82.
Comstock, J. P. and J. R. Ehleringer. 1992. Plant adaptation in the Great Basin and Colorado Plateau. The Great Basin Naturalist 52(3):195-215.
D'Antonio, C. M., and P. M. Vitousek. 1992. Biological invasions by exotic grasses, the grass/fire cycle, and global change. Annual Review of Ecology and Systematics 23:63-87
Daubenmire, R. 1970. Steppe vegetation of Washington. Technical bulletin 62. Washington Agriculture Experiment Station. 131 p.
Daubenmire, R. 1975. Plant succession on abandoned field, and fire influences in a steppe area in southeastern Washington. Northwest Science 49(1):36-48.
Davies, K. W., and D. D. Johnson. 2008. Managing medusahead in the intermountain west is at a critical threshold. Rangelands 30(4):13-15.
Davies, K. W., and T. J. Svejcar. 2008. Comparison of medusahead-invaded and noninvaded Wyoming big sagebrush steppe in southeastern Oregon. Rangeland Ecology and Management 61(6):623-629.
Davies, K. W., A. M. Nafus, and M. D. Madsen. 2013. Medusahead invasion along unimproved roads, animal trails, and random transects. Western North American Naturalist 73(1):54-59.
Davies, K. W., C. S. Boyd, D. D. Johnson, A. M. Nafus, and M. D. Madsen. 2015. Success of seeding native compared with introduced perennial vegetation for revegetating medusahead-invaded sagebrush rangeland. Rangeland Ecology & Management 68(3):224-230.
Dobkin, D. S., and J. D. Sauder. 2004. Shrub steppe landscapes in jeopardy: distributions, abundances, and the uncertain future of birds and small mammals in the Intermountain West. High Desert Ecological Research Institute, Bend, OR.
Dobrowolski, J. P., M. M. Caldwell, and J. H. Richards. 1990. Basin hydrology and plant root systems. Pages 243-292 in C. B. Osmond, L. F. Pitelka, and G. M. Hidy (eds.). Plant biology of the basin and range. Springer-Verlag, New York.
Furbush, P. 1953. Control of Medusa-Head on California Ranges. Journal of Forestry 51(2):118-121.
Furniss, M. M. and W. F. Barr. 1975. Insects affecting important native shrubs of the northwestern United States Gen. Tech. Rep. INT-19. Intermountain Forest and Range Experiment Station, U.S. Department of Agriculture, Forest Service. Ogden, UT. 68 p.
Harris, G. A. 1967. Some Competitive Relationships between Agropyron spicatum and Bromus tectorum. Ecological Monographs 37(2):89-111.
Hironaka, M. 1994. Medusahead: Natural Successor to the Cheatgrass Type in the Northern Great Basin. In: Proceedings of Ecology and Management of Annual Rangelands. USDA Forest Service, Intermountain Research Station. Gen. Tech. Rep. INT-GTR-313. Pages 89-91.
James, J., K. Davies, R. Sheley, and Z. Aanderud. 2008. Linking nitrogen partitioning and species abundance to invasion resistance in the Great Basin. Oecologia 156(3):637-648.
Johnson, B. G., Johnson, D. W., Chambers, J. C., Blank, B. R. 2011. Fire effects on the mobilization and uptake of nitrogen by cheatgrass (Bromus tectorum L.). Plant and Soil 341(1-2):437-445.
Klemmedson, J. O., and J. G. Smith. 1964. Cheatgrass (Bromus Tectorum L.). The Botanical Review 30(2):226-262.
Koniak, S. 1985. Succession in pinyon-juniper woodlands following wildfire in the Great Basin. The Great Basin Naturalist 45(3):556-566.
Kuntz, D. E. 1982. Plant response following spring burning in an Artemisia tridentata subsp. vaseyana/Festuca idahoensis habitat type. Moscow, ID: University of Idaho. 73 p. Thesis.
Lett, M. S., and A. K. Knapp. 2005. Woody plant encroachment and removal in mesic grassland: Production and composition responses of herbaceous vegetation. American Midland Naturalist 153(2):217-231
Mack, R. N., and D. Pyke. 1983. The Demography of Bromus Tectorum: Variation in Time and Space. Journal of Ecology 71(1):69-93.
MacMahon, J. A. 1980. Ecosystems over time: succession and other types of change. In: Waring, R., (ed.) Proceedings—Forests: fresh perspectives from ecosystem analyses. Biological Colloquium. Corvallis, OR: Oregon State University. Pages 27-58.
Mangla, S., R. Sheley, and J. J. James. 2011. Field growth comparisons of invasive alien annual and native perennial grasses in monocultures. Journal of Arid Environments 75(2):206-210.
Miller, H. C., Clausnitzer, D., and Borman, M. M. 1999. Medusahead. In: R. L. Sheley and J. K. Petroff (eds.). Biology and Management of Noxious Rangeland Weeds. Corvallis, OR: Oregon State University Press. Pages 272-281.
Miller, R. F., and Tausch, R. J. 2001. The Role of Fire in Juniper and Pinyon Woodlands: A Descriptive Analysis. In: Galley. K. M. and T. P. Wilson (eds.), Fire Conference 2000: The First National Congress on Fire Ecology, Prevention, and Management; Tallahassee, FL: Tall Timbers Research Station, San Diego, CA, USA. Pages 15-30.
Miller, R. F., J. C. Chambers, D. A. Pyke, F. B. Pierson, and C. J. Williams. 2013. A review of fire effects on vegetation and soils in the Great Basin region: response and ecological site characteristics. Gen. Tech. Rep. RMRS-GTR-308. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station. 126 p.
Monaco, T. A., C. T. Mackown, D. A. Johnson, T. A. Jones, J. M. Norton, J. B. Norton, and M. G. Redinbaugh. 2003. Nitrogen effects on seed germination and seedling growth. Journal of Range Management 56(6):646-653.
Pellant, M., and C. Hall. 1994. Distribution of two exotic grasses in intermountain rangelands: status in 1992. USDA Forest Service Gen. Tech. Rep. INT-GTR-313S. Pages 109-112.
Rice, P. M. 2005. Medusahead (Taeniatherum caput-medusae (L.) Nevski). In: C. L. Duncan and J. K. Clark (eds.). Invasive plants of range and wildlands and their environmental, economic, and societal impacts. Weed Science Society of America, Lawrence, KS.
Sheley, R. L., Svejcar T. J. 2009. Response of bluebunch wheatgrass and medusahead to defoliation. Rangeland Ecology & Management 62(3):278-283.
Sheley, R. L., E. A. Vasquez, A. Chamberlain, and B. S. Smith. 2012. Landscape-scale rehabilitation of medusahead (Taeniatherum caput-medusae)-dominated sagebrush steppe. Invasive Plant Science and Management 5(4):436-442.
Tausch, R. J. 1999. Historic pinyon and juniper woodland development. In: S. B. Monsen and R. Stevens, (comps.). Proceedings: Ecology and management of pinyon-juniper communities within the Interior West. 1997, September 15-18. Provo, UT. Proceedings RMRS-P-9. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Provo, UT. Pages 12-19.
Tisdale, E. W. and M. Hironaka. 1981. The sagebrush-grass region: A review of the ecological literature. Bulletin 33. University of Idaho, Forest, Wildlife and Range Experiment Station. Moscow, ID. 31p.
Vollmer, J. L., and J. G. Vollmer. 2008. Controlling cheatgrass in winter range to restore habitat and endemic fire United States Department of Agriculture, Forest Service. RMRS-P-52. Pages 57-60.
Wagner, J. A., R. E. Delmas, J. A. Young. 2001. 30 years of medusahead: return to fly blown-flat. Rangelands 23(3):6-9.
Whisenant, S., 1999. Repairing Damaged Wildlands: a process-orientated, landscape-scale approach (Vol. 1). Cambridge, UK: Cambridge University Press. 312 p.
Wright, H. A. 1985. Effects of fire on grasses and forbs in sagebrush-grass communities. In: K. D. Sanders and J. Durham, (eds.). Rangeland Fire Effects: A Symposium. 1984, November 27-29. USDI-BLM, Boise, ID. Pages 12-21.
Young, J. A., R. E. Eckert, Jr., and R. A. Evans. 1979. Historical perspectives regarding the sagebrush ecosystem. In: The Sagebrush Ecosystem: A Symposium. 1978, April. College of Natural Resources, Utah State University, Logan, UT. Pages 1-13.
Young, R. P. 1983. Fire as a vegetation management tool in rangelands of the Intermountain region. In: Monsen, S.B. and N. Shaw (eds). Managing Intermountain rangelands—improvement of range and wildlife habitats: Proceedings. 1981, September 15-17; Twin Falls, ID; 1982, June 22-24; Elko, NV. Gen. Tech. Rep. INT-157. Ogden, UT. U.S. Department of Agriculture, Forest Service, Intermountain Forest and Range Experiment Station. Pages 18-31.
Major Land Resource Area
MLRA 023X
Malheur High Plateau
Subclasses
- R023XF082CA–STONY LOAM 9-12"
- R023XF091CA–LOAMY UPLAND 9-12"
- R023XY006NV–LOAMY 8-10 P.Z.
- R023XY019OR–SILT LOAM TERRACE 10-12 PZ - Inactive to 024XY019OR
- R023XY020NV–LOAMY 10-12 P.Z.
- R023XY038NV–DROUGHTY LOAM 8-10 P.Z.
- R023XY039NV–LOAMY SLOPE 10-14 P.Z.
- R023XY040NV–GRANITIC FAN 8-10 P.Z.
- R023XY049NV–GRANITIC SOUTH SLOPE 8-12 P.Z.
- R023XY051NV–SANDY 8-12 P.Z.
- R023XY057NV–GRANITIC LOAM 10-12 P.Z.
- R023XY068NV–GRANITIC LOAM 8-10 P.Z.
- R023XY082NV–LOAMY FAN 10-12 P.Z.
- R023XY097NV–LOAMY FAN 8-10 P.Z.
- R023XY099NV–CHANNERY HILL 8-10 P.Z.
- R023XY101NV–STONY SLOPE 8-10 P.Z.
- R023XY212OR–LOAMY 10-12 PZ
- R023XY220OR–CLAYEY 10-12 PZ
- R023XY222OR–SHALLOW LAVA 10-12 PZ
- R023XY300OR–SOUTH SLOPES 10-12 PZ
- R023XY301OR–DROUGHTY SOUTH SLOPES 11-13 PZ
- R023XY308OR–NORTH SLOPES 10-12 PZ
- R023XY602OR–ARID NORTH 8-10 PZ
- R023XY604OR–ARID PLAINS 8-11 PZ
Correlated Map Unit Components
21659207, 21660403, 21660530, 21659998, 21660004, 21659769, 21659200, 21659158, 21660410, 21659525, 21659810, 21659805, 21660145, 21660217, 21659803, 21660111, 21659872, 21660022, 21659526, 21659801, 21659806, 21659598, 21660137, 21660218, 21660364, 21660505, 21500587, 21500945, 21500894, 21501135, 21501226, 21500368, 21500366, 21501128, 21501348, 21501020, 21500615, 21500360, 21500986, 21500356, 21501079, 21500666, 21500657, 21500956, 21501351, 21500819, 21501409, 21501296, 21500373, 21500805, 21501031, 21501056, 21500658, 21500598, 21501353, 21500797, 21500650, 21501280, 21500238, 21500737, 21500271, 21500848, 21501158, 21501003, 21501047, 21500936, 21501167, 21500803, 21500348, 21501614, 21501228, 21501903, 21501902, 21501059, 21500292, 21500251, 21501166, 21500536, 21500540, 21500532, 21500250, 21500275, 21500610, 21500398, 21500239, 21501612, 21501061, 21501359, 21500267, 21500236, 21500747, 21500735, 21500765, 21501001, 21500758, 21500770, 21501209, 21501223, 21501050, 21500829, 21500980, 21501442, 21501436, 21501278, 21500825, 21501064, 21501401, 21500999, 21501089, 21501094, 21500583, 21501080, 21500818, 21500371, 21501189, 21501219, 21501206, 21500384, 21500801, 21501006, 21501136, 21500641, 21501229, 21500948, 21500755, 21500400, 21500270, 21501023, 21501163, 21500364, 21501285, 21501317, 21500767, 21501322, 21501650, 21500981, 21500593, 21501188, 21501292, 21501313, 21582462, 21582463, 21582211, 21582117, 21581373, 21582206, 21589609, 21590006, 21590101, 21589510, 21589971, 21589753, 21590121, 21589821, 21589532, 21590107, 21589405, 21590123, 21589976, 21590011, 21589846, 21590015, 21589985, 21589944, 21590081, 21589718, 21589730, 21590084, 21589425, 21589914, 21589787, 21589970, 21589538, 21589582, 21589785, 21589939, 21589403, 21589408, 21589719, 21589541, 21589412, 21589714, 21589789, 21589913, 21589421, 21589779, 21590089, 21589699, 21589945, 21589875, 21589547, 21589870, 21590035, 21589625, 21589722, 21589727, 21589924, 21590021, 21589908, 21589444, 21589770, 21590140, 21589957, 21589772, 21589416, 21589921, 21589744, 21589430, 21590027, 21589688, 21589952, 21590285, 21590293, 21590654, 21590648, 21590522, 21590351, 21590554, 21590758, 21590928, 21590947, 21590695, 21590336, 21590847, 21590743, 21590619, 21590749, 21590808, 21590752, 21590600, 21590563, 21590642, 21590751, 21590807, 21590685, 21590324, 21590401, 21590745, 21590742, 21590819, 21590827, 21590347, 21590362, 21590896, 21590630, 21590774, 21590611, 21590616, 21590462, 21590711, 21590412, 21590418, 21590323, 21591670, 21605101, 21604623, 21604299, 21605185, 21604819, 21605109, 21604589, 21604262, 21605157, 21604264, 21605162, 21605114, 21604842, 21604874, 21605164, 21604142, 21604295, 21604917, 21604919, 21604277, 21604431, 21604474, 21604498, 21604508, 21604800, 21605103, 21604815, 21604513, 21604523, 21604526, 21604830, 21604587, 21604849, 21604859, 21605184, 21604831, 21604720, 21604348, 21729585, 21730198, 21730199, 21729561, 21730191, 21729284, 21730194, 21728757, 21730197, 21729584, 21729186, 21729450, 21729437, 21729615, 21729433, 21729628, 21729439, 21729440, 21729287, 21729449, 21730190, 21729560, 21729559, 21729555, 21729554, 21729445, 22168269, 22168243, 22168270, 22175461, 22176721, 22176866, 22175896, 22175098, 22175676, 22176722, 22176865, 22176725, 22176110, 22176709, 22176706, 22176660, 22176664, 22176724, 22175804, 22176705, 22175518, 21604762, 21604585, 21605093, 21605178, 21604910, 21604588, 21604847, 21604854, 21604857, 21604040, 21604245, 21604901, 22176996, 22175009, 22175634, 22176872, 22176530, 22175938, 22176595, 22176134, 22176526, 22176610, 22176873, 22176621, 22176493, 22175202, 22176175, 22176469, 22176465, 22176602, 22176815, 22176374, 22175452, 22176646, 22175589, 22176392, 22176635, 22175946, 22176403, 22176387, 22175886, 22175187, 22176608, 22175171, 22176400, 22176410, 22175226, 22175837, 22176636, 22176630, 22175062, 22175623, 22175965, 22175442, 22175443, 22175057, 22177005, 22176323, 22175370, 22176373, 22176310, 22176622, 22175320, 22175317, 22175150, 22176926, 22175288, 22175276, 22175272, 22176993, 22175537, 22175000, 22175262, 22176237, 22176238, 22175872, 22176209, 22176562, 22176563, 22176184, 22176185, 22176189, 22175230, 22176180, 22176557, 22175891, 22175250, 22176583, 22176617, 22175937, 22176594, 22176133, 22176525, 22175933, 22175951, 22176272, 22176587, 22176404, 22175665, 22175234, 22175144, 22176590, 22176193, 22175890, 22175911, 22175912, 22176892, 22176824, 22175979, 22176204, 22177015, 22177012, 22177610, 22177511, 22177409, 22177151, 22177045, 22177054, 22177056, 22177426, 22177172, 22177057, 22177058, 22177428, 22177429, 22177304, 22177305, 22177431, 22177311, 22177308, 22177307, 22177306, 22177258, 22177027, 22177664, 22177108, 22177210, 22177456, 22177220, 22177024, 22177702, 22177382, 22177383, 22177137, 22177658, 22177612, 22177508, 22177509, 22177438, 22177611, 22177512, 22177152, 22177132, 22177581, 22177583, 22177584, 22177259, 22177588, 22168435, 22168265, 22168248, 22168258, 22170557, 22171301, 22170565, 22170816, 22170477, 22170473, 22171149, 22171260, 22171252, 22171254, 22171255, 22171253, 22170476, 22171235, 22171203, 22170468, 22171153, 22171152, 22171150, 22171146, 22171144, 22171142, 22171139, 22171137, 22171133, 22170898, 22170782, 22171140, 22171128, 22171127, 22171125, 22170991, 22170835, 22171123, 22171122, 22170784, 22170976, 22170554, 22170469, 22170467, 22170464, 22170463, 22170459, 22170456, 22170455, 22170944, 22170942, 22170943, 22170934, 22170932, 22170933, 22170551, 22170864, 22170817, 22170564, 22170815, 22170811, 22170804, 22170802, 22170800, 22170466, 22170785, 22170783, 22170781, 22171302, 22170780, 22170481, 22170710, 22170707, 22170662, 22170474, 22170660, 22170627, 22170658
Stage
Provisional
Contributors
T Stringham (UNR under contract with BLM)
DMP
Click on box and path labels to scroll to the respective text.